Today the FDA announced that it had declined to approve Ocaliva for treatment of NASH. Intercept Pharmaceuticals was set to be the first FDA approved NASH treatment, despite relatively mediocre Phase 3 Results announced in late 2019. This is a major disappointment for the tens of millions of NASH patients that were hoping for an imminent treatment to be available.
Continue reading “FDA declines to approve Intercept’s Ocaliva”Tag: NASH
Genfit Phase III NASH trial ends in failure
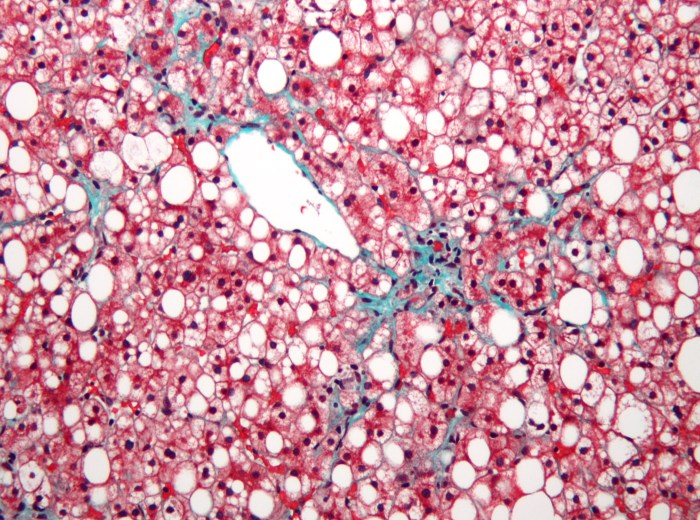
Last night French biotech company Genfit released the results of their widely anticipated Phase III study on Elafibranor, a NASH therapeutic that had shown tremendous promise in earlier trials.
Unfortunately, the trial failed to meet both the primary and secondary endpoints. The treated patients response wasn’t statistically meaningful versus the placebo control group. Genfit’s CEO Pascal Prigent said “These results are highly disappointing.” The stock is down by over 65% today.
This setback leaves us with just Abbvie’s cenicriviroc study to look forward to this year, due in late 2020 if there are no delays due to the Covid crisis. Intercept’s Ocaliva is due for FDA approval soon, but seems to have limited effectiveness in specific NASH cases.
The best treatment for NASH and NAFLD remains a healthy diet and lifetsytle changes for the foreseeable future.
The Best Diagnostic Tool for NASH
One of the questions I see asked all of the time is what kind of testing is the best to determine if someone has NASH. I’ve written about the challenge of diagnosis before, but there are a lot of scared individuals out there trying to get answers from their under-educated primary case physicians, and everyone wants to skip the preliminary steps and go straight to a definitive diagnosis. If only it were that easy!
Continue reading “The Best Diagnostic Tool for NASH”Watch the ALF NASH Day Webinar I participated in

For the 2nd International NASH Day yesterday I took part in a webinar along with Dr. Tuan Pham of the University of Utah. Dr. Pham gave a great overview of the latest in the NASH space and I shared the story of my mom to provide an advocate perspective.
You can view the video here:
“Give It Up” for International NASH Day!
For #NASHDAY I’m asking everyone I know to “Give It Up” for NASH Awareness. It could be sugar, coffee, alcohol, TV, politics…literally anything! Write about what you are giving up on the sign attached and share it all over social media next Wednesday June 12th to help spread the word.
And if you can take the $5 that would have gotten you a cup of coffee and instead donate it to the American Liver Foundation’s NASH Day page, you can help raise awareness, provide education and support, and help fund treatments for NASH patients in the future.
Hope to see you all Giving It Up on June 12th!
Visit https://alfmad.org/greaternewyork/support/#NASHday to donate.!

My American Liver Foundation NASH Advocacy Video
A few months ago I was honored to take part in an American Liver Foundation effort to educate and raise awareness for NASH. I travelled to University of San Diego’s Altman Clincial and Translational Research Institute where I recorded a video telling my mom’s story so that others could learn and benefit from our loss. I was joined by a panel of individuals currently living with various stages of NASH and a few distinquished medical experts in the field.

The videos are finally being released as part of our leadup to the 2nd International NASH Day on June 12th. You can watch the entire video below.
For #NASHDAY I’m asking everyone I know to “Give It Up” for NASH Awareness. It could be sugar, coffee, alcohol, TV…literally anything. Post about what you are giving up on social media to help spread the word. And if you can take the $5 that would have gotten you a cup of coffee and instead donate it to the American Liver Foundation’s NASH Day page, you can help raise awareness, provide education and support, and help fund treatments for NASH patients in the future.
https://alfmad.org/greaternewyork/support/#NASHday to donate.
Intercept Pharma announces successful Phase 3 NASH trials
Will move for approval in US and Europe later this year
Eagerly awaited Phase 3 trial results were released early Tuesday morning from Intercept Pharmaceuticals, showing positive results for the study’s primary goal of showing a statistically significant reduction in liver fibrosis compared to placebo. As a result, Intercept will seek approval in the US and Europe to permit the drug, known as Ocaliva, to be used as the first ever treatment for NASH-related fibrosis. The stock price rose as high as 23% on the news.
Continue reading “Intercept Pharma announces successful Phase 3 NASH trials”Our Sugar Addiction is Slowly Killing Us

I have a friend who once lost a whole bunch of weight by removing two simple things from his diet. Cheese and soda. This was well before the current Paleo diet craze, when sugary drink taxes weren’t even whispered in the most progressive of city councils. It was also well before I knew anything about the liver or the difference between sucrose and fructose in the sugar I consumed. His story always stuck with me, though I never realized why it had worked.
Ever since the untimely death of my mother from NASH, I have spent a lot of time learning about the underlying causes that likely led to her liver failure. Along the way, I had to reexamine a number of misconceptions and prejudicies I’ve had about health and eating. Whether from genetics or pure luck, I’ve been blessed all my life with never having to worry about weight or what I eat. I never put a lot of thought about what I was putting inside my body until i started to read research about how obesity and metabolic syndrome developed, and why we were suddenly facing a worsening crisis over the last 50 years.
What I’ve found has changed my entire view on food & health.
The link between Gallbladder Disease and NASH
When my mom passed away at 62 years old six weeks after being diagnosed with a disease I’d never heard of before, it sent me searching for answers. NASH is still widely unknown and the etiology is not well understood. Yes, it is largely linked to obesity, but there are many other risk factors that seem to be playing a role. One such factor that I discovered last year was the link between hypothyroidism and NASH, which my mom had for decades.
Recently I have been working on some projects on how the liver works, and found myself intrigued when writing about the function of the gallbladder. For those that are unaware, its main function is storing bile to be used in the digestion of fats. As I wrote about it, alarm bells began ringing in my head. NASH is the end stage of fatty liver disease, caused by an excess buildup of fat in the liver.
Gallbladder surgery (known as cholecystectomy) is one of the most common surgical procedures in the world, with over 600,000 procedures performed annually in the USA. It is considered completely safe with no long-term negative effects. My mom had her gallbladder removed years ago after suffering gallstones, as is the standard medically recommended procedure. I wondered if there was any studies on possible links between it and NASH, and began searching.
The answer is a resounding yes. Not only is there a strong correlation between gallbladder disease and NASH in several studies, but there is an even stronger independent link between cholecystectomy and NAFLD/NASH.
Continue reading “The link between Gallbladder Disease and NASH”
Genfit and LabCorp team up to develop NASH diagnostic tests

One of the most difficult challenges with NASH is that it is so difficult to diagnose. While many Biotech companies are working hard to develop a cure for NASH, some are also busy developing new innovative methods to detect NASH without requiring an invasive liver biopsy.
Genfit is one of the few companies due to release Phase 3 trials of NASH treatments in 2019. Not content with focusing merely on a cure, they are also nearing completion of trials on a new blood test for earlier detection of NASH. They have just recently announced a partnership with LabCorp, which will allow them access to a much wider audience for the clinical research required to validate the new test.
Continue reading “Genfit and LabCorp team up to develop NASH diagnostic tests”

