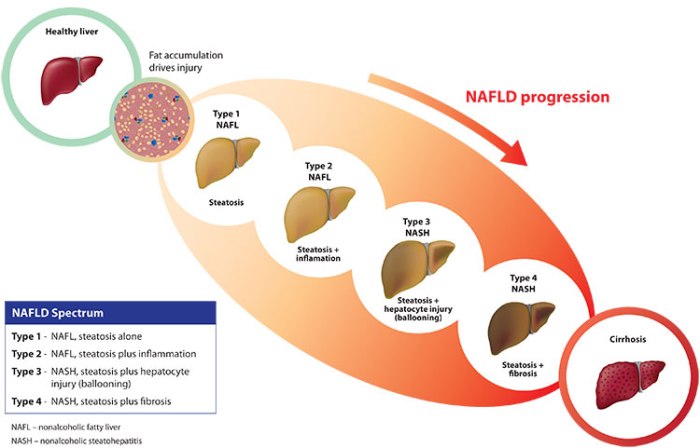
NASH is the acronym for nonalcoholic steatohepatitis. It’s a type of advanced liver disease that is clinically indistinguishable from alcohol-induced liver damage, but whose causes are totally different. Late stage NASH leads invariably to chronic cirrhosis and ultimately liver failure and death. The most common cause of NASH is long-term obesity, and so the threat has grown in relation to the rising rates of obesity in western society. Therefore NASH is quickly becoming an epidemic, with estimates of 15 to 30 million Americans living with the disease, the vast majority of whom are undiagnosed. They are living with a ticking time bomb inside their bodies. Continue reading “What is NASH?”
Wegovy Receives Accelerated FDA Approval To Treat MASH
For many years doctors have told me that the best treatment for NAFLD/NASH (now referred to medically as MAFLD/MASH) was simply losing weight. That’s because we did not have any pharmaceutical treatments (until ezdiffra (resmetirom) from Madrigal in 2024), and until late stage fibrosis & cirrhosis develops, most liver disease is fully reversible.
On August 15, 2025, the FDA granted accelerated approval to Wegovy (semaglutide 2.4 mg) for treating adults with non-cirrhotic MASH and moderate to advanced liver fibrosis. This is an enormous milestone in the treatment of this disease. This makes Wegovy the first GLP‑1 receptor agonist to earn that distinction, and only the second overall drug to treat MASH.
GLP-1 drugs have obviously been all the rage for weight loss, and there has been some off-label use of them to treat liver disease like MASH, but in the Phase III ESSENCE trial, after 72 weeks, 63% of patients on Wegovy saw their steatohepatitis resolved without any worsening of fibrosis, compared to just 34% with placebo. And 37% saw an improvement in fibrosis with no worsening of inflammation, versus 22% in the placebo group. So it is not just helping patients lose weight but showing meaningful improvement for liver disease.
Dr. Martin Holst Lange, Novo Nordisk’s Chief Scientific Officer, put it bluntly: “Wegovy is now uniquely positioned as the first and only GLP‑1 treatment approved for MASH, complementing the already proven weight loss, cardiovascular benefits and extensive body of evidence linked to semaglutide”
Living with liver disease will still require lifestyle changes; these are foundational to the process. But now, there’s medicine that can resolve inflammation and heal fibrosis, beyond just nudging the scale downward. A tremendous step forward!
Our healthcare system is failing us

The energy in the room was electric. Packed to standing-room-only capacity, it held diverse representatives from nations as far afield as Qatar, Africa, and Brazil. These were not just academics or clinicians—they were heavy hitters in global health, some of whom had spent decades shaping the fight against diseases like HIV, malaria, and obesity. And yet, amidst this impressive gathering, a dark truth emerged: our healthcare system is failing millions of people with fatty liver diseases, including Non-Alcoholic Fatty Liver Disease (NAFLD) and its more severe form, Non-Alcoholic Steatohepatitis (NASH).
Dr. Christoph Benn, an influential early leader of the world-renowned Global Fund, did not mince words. During the discussion, he bluntly stated that until invited to this event, he’d never heard of NAFLD or NASH—not by these names, nor by their newer terminology, MASLD and MASH. His message was painfully clear: if a disease impacts an estimated 100 million Americans and remains largely invisible on the global stage, then the system designed to promote awareness and action has failed. What’s more, the 2024 Lancet publication on global health—an agenda-setting document for the next decade that for the first time is making a major shift from infectious diseases to non-communicable diseases (NCDs) —makes no mention of liver disease at all. Zero.
Let that sink in. Our healthcare policymakers are flying blind on an epidemic of massive proportions.
“100 million Americans are impacted by NAFLD, yet it remains invisible on the global stage.”
It was late September 2024, and for the first time in history there was a United Nations General Assembly (UNGA) sidelines event focused on liver disease, specifically MAFLD/MASH. UNGA Week is an important series of side events that include panel discussions, focus groups, and gatherings of global leaders and experts, often taking place in parallel to the formal UNGA sessions and are organized by UN member states, UN agencies, non-governmental organizations (NGOs), and private sector groups. These UNGA sidelines are key opportunities for advocacy, coalition-building, and policy discussions on pressing global issues like health, climate change, education, and sustainable development.


I have attended many NASH-related conferences and panel discussions in the years since I started NASHAWARE and began working with ALF, but they were also from a US-centric standpoint, and so the global viewpoints that were expressed during this amazing UNGA session hosted by The Economist Impact were refreshing and invigorating. The real eye-opener for me was how far we really have to go to make an impact in global health policy. You may have thought that with NASH drugs being a hot multi-billion dollar industry that there would be at least some action on the policy front, but other than ALF’s recent success in helping secure funding for the first US government-funded NAFLD study there really hasn’t been anything of note, and as the participants pointed out, plenty of missed opportunities.
The stakes have ever been higher or more clear, as illustrated by a sobering statistic from Dr. Arun Sanyal. “When a person shows up a emergency room for the first time with a complication of cirrhosis, two out of three individuals, it is the first day their liver disease has been diagnosed.” This is not just an oversight; it’s a systemic failure—a glaring indictment of our approach to prevention, early detection, and education. As Dr. Jeffrey Lazarus put it: “For everyone working in public health, global health, even health economics…it’s not that we’re not on their agenda. Some of them don’t even know this disease exists.”
For 20 or more years, the entire healthcare system has failed that patient.” – Dr. Arun Sanyal
Globally, countries like Qatar offer a blueprint for action. Their Minister of Health, Her Excellency Al Kuwari, spoke about how liver testing has been integrated into national health screenings. By embedding obesity and diabetes monitoring into the fight against liver disease, Qatar has not only halted the growth of obesity but also set a bold target to reduce it by 8% by 2030. “Yes we need to continue to study this, but we have enough data to take action” she declared. While they have the benefit of a small and affluent population, they can still inspire other nations through action.
Johanna Ralston, CEO of the World Obesity Foundation, made the observation that NAFLD is both “everywhere and nowhere.” Her point was clear: the disease is ubiquitous, affecting millions, yet remains absent from critical discussions on high-priority non-communicable diseases by organizations like the WHO. “It’s an exciting time for all of us working in this metabolic health space,” she mentioned, highlighting the ways that all of the various disease communities are coming together. “The timing is now,” Dr Sanyal added, echoing the call for action and collaboration to combat this invisible epidemic.
Contrast this with the fragmented and often lethargic approach in the U.S., where red tape, special interests, and stigma hinder progress. For instance, stigma around liver diseases being linked to alcohol persists, leading to misconceptions even within families. This stigma discourages both patients and policymakers from engaging in meaningful discussions, let alone action. We are also facing an uphill battle on messaging, which is hindered by a lack of clear and concise messages, even around the basic facts like what to call it (several panelists misstated the new terminology). Advocacy groups like the American Liver Foundation are pushing against this tide but are hindered by limited budgets and an uphill battle for donor dollars.
So, what’s the solution? The answer lies in disruptive advocacy—moving beyond just raising awareness to shaking the very foundations of our healthcare and political systems. As a former UK NHS legal counsel argued at the event, awareness campaigns often fall victim to shifting political winds. Instead, we must demand accountability by presenting stark, undeniable truths: “The healthcare system is failing millions of Americans. Here are the X lives lost and Y economic costs of inaction. Here’s what we need to fix it, and here’s how politicians can be remembered for saving lives, not abandoning them.”
It’s also about partnership and focus. We need to stop seeing liver disease in isolation and address the elephant in the room: metabolic dysfunction. NAFLD and NASH are intrinsically tied to obesity, diabetes, and poor nutrition. Advocacy must break through silos and unite stakeholders across fields—from obesity-focused NGOs to diabetes organizations—to tackle the root causes. As one speaker put it, we need to see “the elephant in the room, not the individual trunk or legs.”

The medical community must also adapt its approach to address the growing burden of NAFLD and NASH. Overspecialization within healthcare often creates silos that hinder a comprehensive approach to these diseases. Specialists in hepatology, gastroenterology, endocrinology, and cardiology need to collaborate more effectively, sharing knowledge and resources to tackle the interconnected nature of metabolic dysfunction. Medical training and research funding must emphasize cross-disciplinary education and solutions, ensuring that future healthcare providers are equipped to address these complex, multifaceted conditions.
Yet even the most compelling arguments won’t resonate without clarity of messaging. Attendees mentioned using visuals that elicit emotional responses; imagine a basketball court: ten kids playing a game. Now imagine telling those watching that one of these kids will develop MASLD or MASH before adulthood. One in ten. That’s the kind of visceral imagery we need to cut through the noise and make statistics real.
And then there’s the power of technology. A young doctor from Africa highlighted how AI tools could amplify these efforts, from translating advocacy materials into multiple languages to identifying high-risk individuals. These tools are there; we just need the vision and will to use them.
The harsh reality is that the healthcare system—both in the U.S. and globally—has left millions to fall through the cracks. But we can change this narrative. By simplifying our message, demanding specific actions, and rallying patients, advocates, and healthcare professionals to disrupt the status quo, we can force the system to do better.
Because the stakes are too high, and the cost of inaction is measured in lives lost—a price we cannot afford to keep paying.
(The entire very informative event can be viewed here.)
Liverfast offers a quick and non-invasive blood test to identify nafld/nash

One of the biggest challenges in tackling the NAFLD/NASH epidemic is that there has traditionally been no easy non-invasive way to detect it in patients. This is a large reason why there are tens of millions of people in the United States alone that are impacted but do not even know it.
That has changed significantly in the last year as a company called Fibronastics has released widely a simple blood screening test that can be used to detect levels of steatosis, inflammation activity, and fibrosis. The LIVERFASt test uses a machine learning algorithm to examine 10 separate biomarkers from the blood draw and uses a scoring system to provide results which normally cannot be identified without a biopsy.
This means that physicians now have a powerful non-invasive tool to help screen for liver illnesses or gauge the severity of diagnosed liver conditions without biopsy (huge!). Fibronastics has recently announced a partnership with The American Liver Foundation to provide free LIVERFASt tests for individuals who qualify, and most insurance companies as well as Medicare will now cover the tests as well.
The accuracy of the test has been well established with medical studies going back to 2019 concluding that it is both “reliable and reproducible”. The scoring system is easy-to-understand and arms patients with clear & concise information to utilize over the course of their journey with liver disease.

“It is a reliable, and reproducible tool which provides grading or staging of the three elementary features of NASH: steatosis, inflammatory activity and fibrosis”
As biopharma continues to search for effective medical treatments, LIVERFASt will become a critical tool in the early detection of NAFLD & NASH.
The Covid-19 Vaccine & Liver Disease
Many people living with Liver Disease have questions about the Covid-19 vaccine, including how effective it is and if there are any risks associated with it specifically related to Liver Disease. This article will attempt to summarize some of the publicly available information on vaccine efficacy and safety for those with Liver Disease. As always, you should consult with your doctor if you have specific questions or concerns.
Continue reading “The Covid-19 Vaccine & Liver Disease”FDA declines to approve Intercept’s Ocaliva
Today the FDA announced that it had declined to approve Ocaliva for treatment of NASH. Intercept Pharmaceuticals was set to be the first FDA approved NASH treatment, despite relatively mediocre Phase 3 Results announced in late 2019. This is a major disappointment for the tens of millions of NASH patients that were hoping for an imminent treatment to be available.
Continue reading “FDA declines to approve Intercept’s Ocaliva”Obesity is a Major Risk Factor in Covid-19 Cases – Perhaps the Largest

Strong evidence that obesity is a major risk factor in Covid-19 severity and poses an increased risk of death has now been demonstrated across multiple studies around the world. The evidence all points to obesity as the preexisting condition with the largest impact on unfavorable outcomes, across all age groups.
The data is especially striking for younger adults, long thought to be at a much lower risk for serious illness from Covid-19. One NYU Langone study of 3,615 Covid patients under 60 years of age showed that “Patients with a BMI of 30-34 were twice as likely to get admitted to the hospital or to be admitted to acute care. Patients with a BMI of 35 or higher were twice as likely to be admitted to the hospital, and three times as likely to end up in the intensive care unit.”
A CDC study found that of its patients, “89% had at least one underlying condition, with obesity being the most common for those between 18 and 64.” A Chinese study showed that obesity tripled the risk of a severe case versus those of normal weights.
Data from UK hospitalizations is even more stark, showing that obesity and metabolic syndrome are associated with a 10 times increase in death. In an article entitled “Covid 19 and the elephant in the room”, Dr. Aseem Malhotra states it bluntly; “OBESITY, THE REAL KILLER BEHIND COVID”
72.7% of patients admitted to ICU are overweight or obese and that those with the related metabolic syndrome have a tenfold increase in mortality from the virus.
Dr. Asseem Malhotra, NHS
Data from a set of studies in France recently published in The Lancet looked at the percentage of ICU patients with Covid that were obese versus the percentage of non-Covid ICU patients. It found a clearly higher rate of ICU admissions among obese patients for Covid vs other causes. This research is also anecdotally supported out of the NYC hospital data.
This blog has discussed the Obesity Epidemic many times before, and I’ve started examining some of the causes of it that are easier to control for. It was always clear that obesity & metabolic syndrome are important for far more than NASH, but this pandemic should serve as a clear wake-up call for the tens of millions of obese individuals around the world. We cannot hide from our health behind the well-intentioned shield of the Body Positivity movement. The warnings are now being shouted loud and clear:
“[P]eople with a body mass index of 30 or more should be taking the same precautions as someone over 65, regardless of their age.”
Dr. Carlos Galvani, Tulane Bariatric Center
Genfit Phase III NASH trial ends in failure
Last night French biotech company Genfit released the results of their widely anticipated Phase III study on Elafibranor, a NASH therapeutic that had shown tremendous promise in earlier trials.
Unfortunately, the trial failed to meet both the primary and secondary endpoints. The treated patients response wasn’t statistically meaningful versus the placebo control group. Genfit’s CEO Pascal Prigent said “These results are highly disappointing.” The stock is down by over 65% today.
This setback leaves us with just Abbvie’s cenicriviroc study to look forward to this year, due in late 2020 if there are no delays due to the Covid crisis. Intercept’s Ocaliva is due for FDA approval soon, but seems to have limited effectiveness in specific NASH cases.
The best treatment for NASH and NAFLD remains a healthy diet and lifetsytle changes for the foreseeable future.
NASH: 2020 Outlook

Around this time last year there were a number of news articles declaring 2019 “The Year of NASH” . Four companies were due to release Phase 3 trial results. Optimism was surging and awareness was beginning to catch on. So how did 2019 turn out, and what does the outlook for NASH in 2020 look like?
A year later:
- Gilead had several Phase 3 studies end that failed to live up to expectations, eliminating Selonsertib as a serious treatment option
- Intercept released mixed results for Ocaliva, but is pushing ahead for FDA Approval. If granted, it would be the first approved NASH treatement. Decision on approval should come after April 2020.
- Abbvie (acquired Allergan) pushed their Phase 3 results for cenicriviroc from 2019 to late 2020.
- Genfit delayed Phase 3 elafibranor results to Q1 2020. It remains a promising undercard.
So while we didn’t get any truly amazing results for treatment in 2019, we did make progress. The 2nd International NASH Day helped reach people across the globe. Awareness has been steadily growing.
And while we have yet to hear from Genfit on their treatment data, they did recently announce tremendeous progress in their quest for a non-invasive diagnostic test. Using a unique 4-biomarker algorithm they have been able to accurately identify NASH and significant fibrosis. Genfit plans to file for FDA approval of the test in 2020.
A reliable non-invasive test for NASH is in some ways even more important than a medical treatment. The danger from NASH comes from years of undiagnosed progression, and we already know that the liver damage NASH can cause is reversible in earlier stages of the disease.
The Stigma of Liver Disease
I do a lot of volunteer work with the American Liver Foundation. This frequently puts me in social settings at food & drink events where, upon hearing the name of the organization, I’m immediately confronted with the biggest stigma of Liver Disease. “Isn’t it a little odd to be talking about liver disease while holding a beer?”
I love these conversations, because the juxtaposition creates a teachable moment that is more likely than not to be remembered.
My response blows up their preconceptions. “Actually, there are over one-hundred types of liver disease that have nothing to do with alcohol. NASH is a serious progressive liver disease affecting over twenty million American’s alone”.
Continue reading “The Stigma of Liver Disease”The Best Diagnostic Tool for NASH
One of the questions I see asked all of the time is what kind of testing is the best to determine if someone has NASH. I’ve written about the challenge of diagnosis before, but there are a lot of scared individuals out there trying to get answers from their under-educated primary case physicians, and everyone wants to skip the preliminary steps and go straight to a definitive diagnosis. If only it were that easy!
Continue reading “The Best Diagnostic Tool for NASH”